As Tom Zoellner writes in Uranium: War, Energy, and the Rock That Shaped the World, "A uranium atom is so overloaded that it has begun to cast off pieces of itself, as a deluded man might tear off his clothes. Before Ernest Rutherford and Frederick Soddy discovered this trait around 1901, the notion of transforming one element into another was thought to be solely the territory of alchemists. Uranium decays into other elements, shedding protons to become protactinium, radium, radon, polonium, and on for a total of 14 transitions, all of them radioactive, until it finds a resting point as lead. ITS TRANSFORMATIONS PROVED THE ALCHEMISTS RIGHT … A LITTLE. He left uranyl potassium sulfate, a type of salt, on a photographic plate in a drawer, and found the uranium had fogged the glass like exposure to sunlight would have. He named it after the recently discovered planet Uranus.įrench physicist Henri Becquerel discovered uranium's radioactive properties-and radioactivity itself-in 1896. It generally appeared where the silver vein ran out, earning it the nickname pechblende, meaning "bad luck rock." In 1789, Martin Klaproth, a German chemist analyzing mineral samples from the mines, heated it and isolated a "strange kind of half-metal"-uranium dioxide. Uranium was first found in silver mines in the 1500s in what's now the Czech Republic. Were it employed that way now, sailing into port could set off defense systems. That weight once compelled shipbuilders to use spent uranium as ballast in ship keels. With a nucleus packed with 92 protons, uranium is the heaviest of the elements. IT'S THE HEAVIEST NATURALLY OCCURRING ELEMENT IN THE UNIVERSE. In the first half of the 20th century, scientists began investigating uranium's innate potential as an energy source, and it has earned its place among the substances that define the "Atomic Age," the era in which we still live. 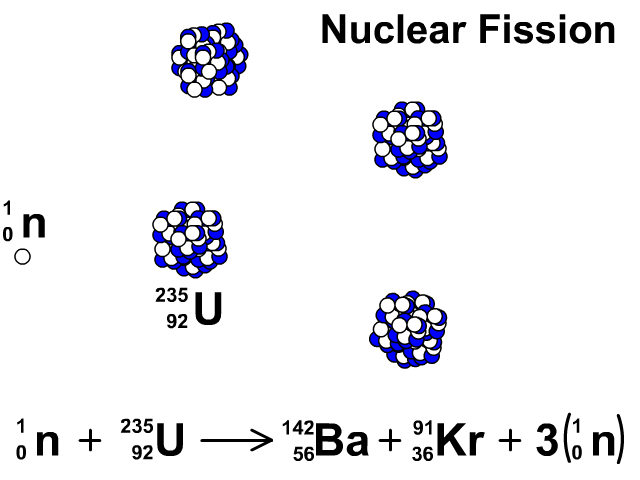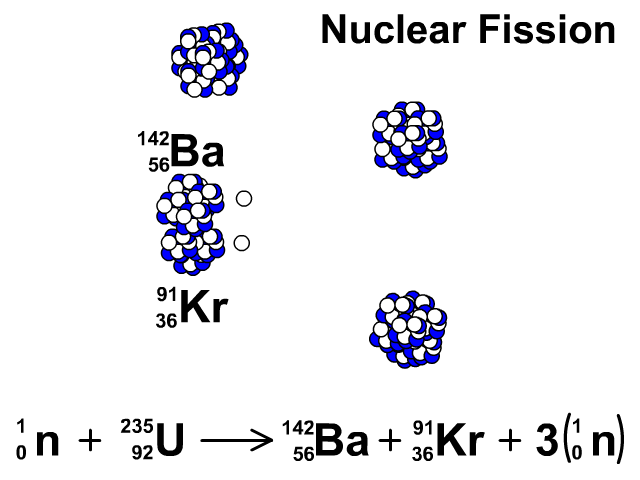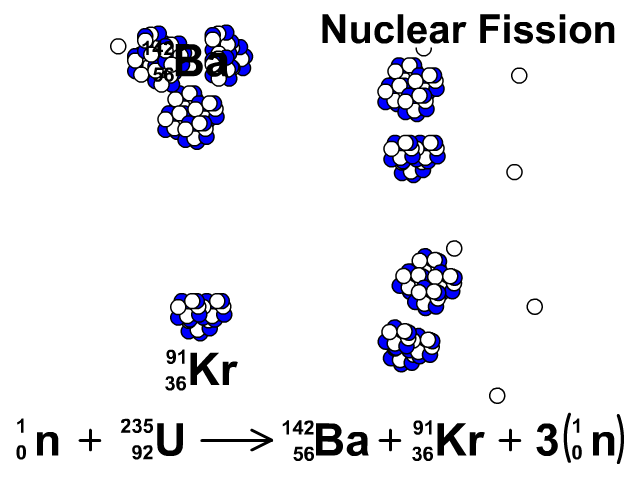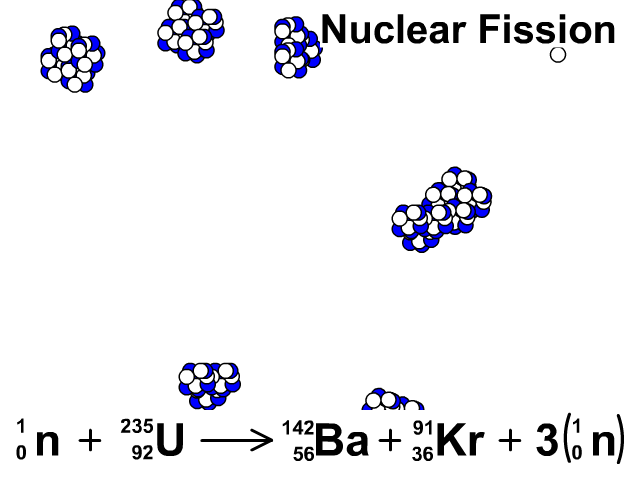
After formal discovery of the element in the late 18th century, it found a useful niche coloring glass and dinner plates. 
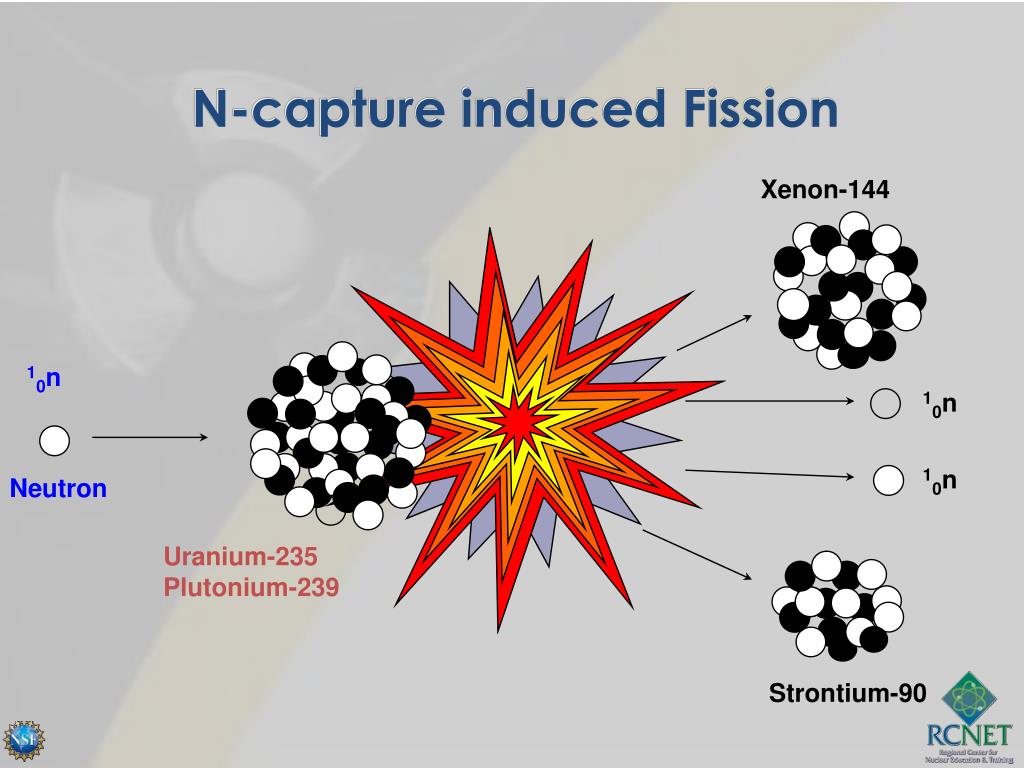
For centuries, heaps of it languished in waste rock piles near European mines. This is the principle how fission fragments heat up fuel in the reactor core.How well do you know the periodic table? Our series The Elements explores the fundamental building blocks of the observable universe-and their relevance to your life-one by one. The positive ions and free electrons created by the passage of the charged fission fragment will then reunite, releasing energy in the form of heat (e.g., vibrational energy or rotational energy of atoms). Creation of ion pairs requires energy, which is lost from the kinetic energy of the charged fission fragment causing it to decelerate. The fission fragments interact strongly with the surrounding atoms or molecules traveling at high speed, causing them to ionize. On the other hand most of the energy released by one fission (~170MeV of total ~200MeV) appears as kinetic energy of these fission fragments. Therefore part of the released energy is radiated away from the reactor (See also: Reactor antineutrinos). Most of the fission fragments are highly unstable (radioactive) nuclei and undergo further radioactive decays to stabilize itself. It is much more probable to break up into unequal fragments, and the most probable fragment masses are around mass 95 (Krypton) and 137 (Barium). The average of the fragment atomic mass is about 118, but very few fragments near that average are found. Typically, when uranium 235 nucleus undergoes fission, the nucleus splits into two smaller nuclei (triple fission can also rarely occur), along with a few neutrons (the average is 2.43 neutrons per fission by thermal neutron) and release of energy in the form of heat and gamma rays. About 85% of all absorption reactions result in fission. Therefore about 15% of all absorption reactions result in radiative capture of neutrons. The cross-section for radiative capture for thermal neutrons is about 99 barns (for 0.0253 eV neutron). Most absorption reactions result in fission reaction, but a minority results in radiative capture forming 236U. For fast neutrons, its fission cross-section is on the order of barns. Uranium 235 is a fissile isotope, and its fission cross-section for thermal neutrons is about 585 barns (for 0.0253 eV neutron).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
